
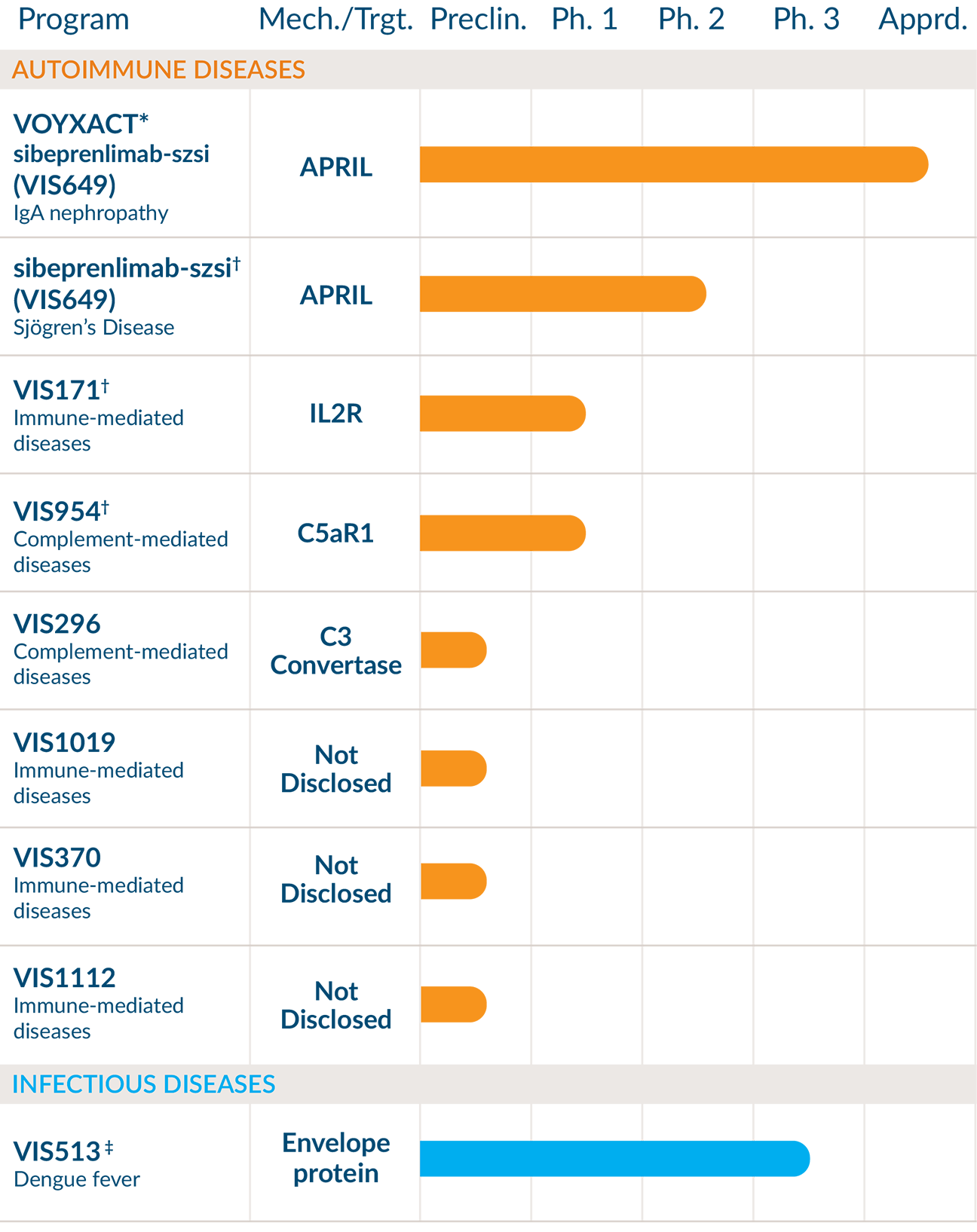
*Marketing and commercialization conducted by Otsuka America Pharmaceutical, Inc., a wholly owned subsidiary of Otsuka Pharmaceutical Co., Ltd. VOYXACT (sibeprenlimab-szsi), a humanized monoclonal antibody that binds to and blocks APRIL, received accelerated approval by the U.S. Food and Drug Administration for the reduction of proteinuria in adults with Primary Immunoglobulin A Nephropathy (IgAN) at risk for disease progression. It has not been established whether VOYXACT slows kidney function decline over the long-term in patients with IgAN. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory clinical trial from the ongoing Phase 3 VISIONARY study evaluating whether VOYXACT slows disease progression as measured by estimated glomerular filtration rate (eGFR) decline at 24 months.
†Clinical development conducted by Otsuka Pharmaceutical and Development, Inc., a wholly owned subsidiary of Otsuka Pharmaceutical Co., Ltd.
‡Clinical development conducted by Serum Institute of India Pvt. Ltd.




